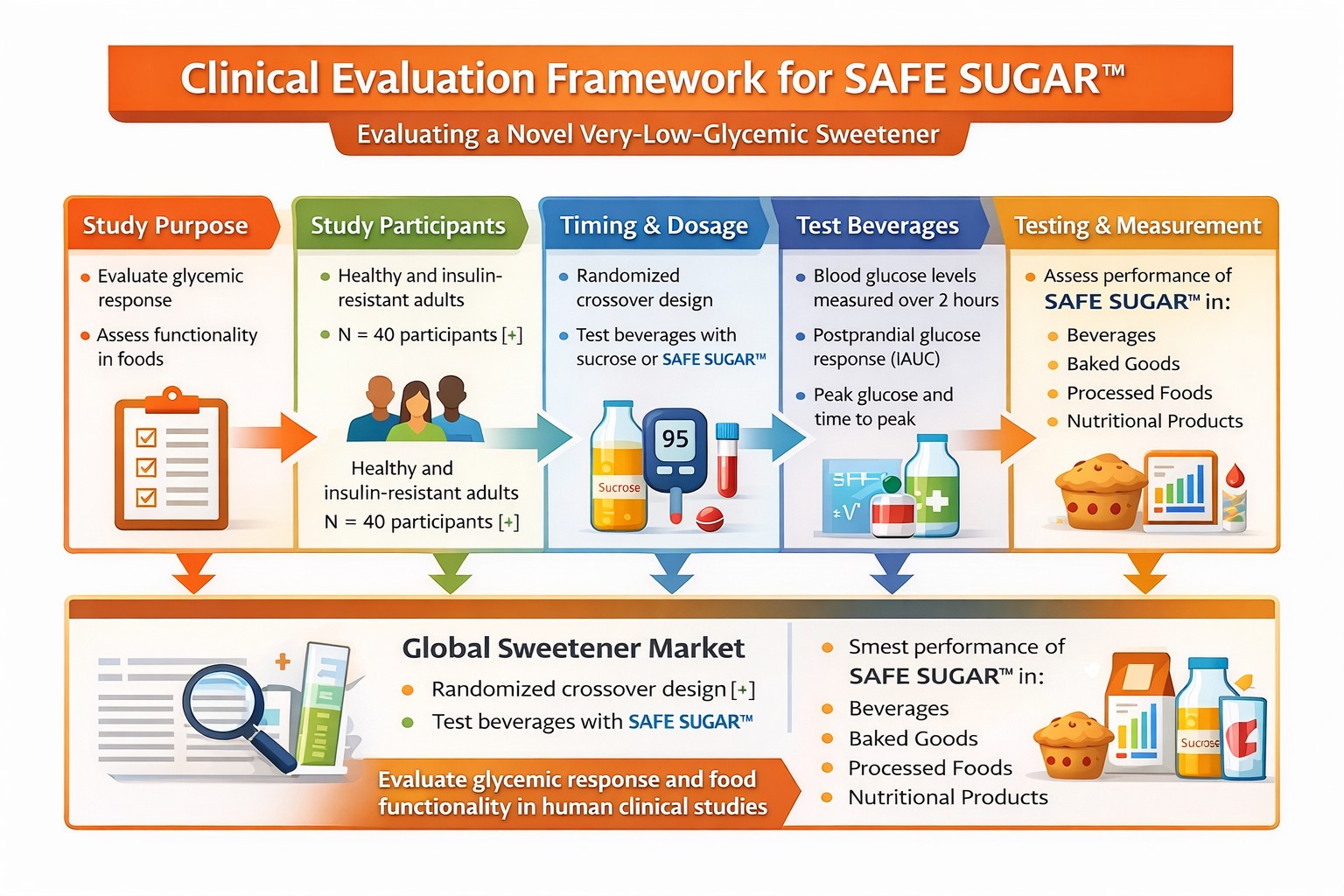
Scientific Validation Strategy
SAFE SUGAR™ is being developed as a very-low-glycemic sweetener platform designed to provide sweetness and functional performance comparable to traditional sugar while reducing post-prandial glucose response.
The validation strategy focuses on evaluating metabolic response, glycemic impact, and functional performance in real food systems.
Clinical Evaluation Framework

Study Design
The evaluation framework is based on a randomized crossover design comparing traditional sucrose with SAFE SUGAR™ in human participants.
Blood glucose levels are measured over a two-hour post-prandial period to determine peak glucose levels, time-to-peak glucose, and incremental area under the curve (iAUC).
Functional Food Testing
SAFE SUGAR™ is evaluated in multiple product categories including beverages, baked goods, processed foods, and nutritional products to assess sweetness performance and manufacturing compatibility.
Expected Outcomes
• Reduced post-prandial glucose response
• Improved glycemic stability compared to traditional sugar
• Functional compatibility with common food manufacturing processes
• Potential integration into diabetic-friendly and metabolic-health product lines
Future Clinical Research
Future studies may include extended metabolic trials, larger population studies, and evaluation in individuals with insulin resistance or Type 2 diabetes.